Long-Term Meloxicam: Protecting Your Heart and Kidneys
By Frankie Sze | 5/2/2026
Beyond the Black Box: Why the Warnings Exist
When meloxicam (Mobic) first entered the market, it was touted for its potential as a more stomach-friendly alternative to traditional, non-selective NSAIDs like ibuprofen. However, the subsequent FDA-mandated class-wide Black Box warnings for all non-aspirin NSAIDs—highlighting significant cardiovascular and gastrointestinal risks—have often left patients feeling apprehensive. As Frankie Sze, I want to clarify that these warnings are not designed to serve as deterrents, but as essential guardrails to ensure medication is used under strict clinical supervision. Understanding these risks is part of what we cover in our Meloxicam 101: Your Daily Guide to Steady Joint Relief.
The complexity of meloxicam lies in its pharmacological footprint; while it is preferentially selective for COX-2, it still inhibits COX-1 to a degree, which contributes to its unique safety profile. Despite this, experts note that “Meloxicam, as a COX-2 preferential NSAID, carries significant cardiovascular risk that increases with COX-2 selectivity and is amplified in patients with established cardiovascular disease.” Interestingly, recent real-world evidence from 2020 to 2024 is nuanced: in a large Korean cohort, the adjusted hazard ratio for thromboembolic events was roughly 3 for meloxicam, compared to 10 for naproxen in post-MI patients, suggesting that our understanding of its safety in high-risk populations is evolving.
Because meloxicam has a long half-life, it provides consistent therapeutic relief, but this extended presence in the bloodstream necessitates vigilant monitoring. In susceptible patients, NSAIDs can induce a rapid fall in glomerular filtration rate (GFR) within hours or days. Consequently, standard care mandates renal function monitoring at baseline, 1-2 weeks post-initiation, and monthly thereafter for the first two months. By adhering to the standard daily dosage range of 7.5 mg to 15 mg and embracing the future of personalized medicine—such as genetic metabolic profiling and home blood pressure monitoring—we can transform these safety warnings from sources of fear into tools for precise, life-sustaining care.

The Heart of the Matter: How Meloxicam Influences Blood Pressure
While meloxicam (Mobic) was initially marketed as a more stomach-friendly alternative to non-selective NSAIDs like ibuprofen, its influence on the cardiovascular system extends far beyond the gastrointestinal tract. Meloxicam is preferentially selective for COX-2 but still retains enough COX-1 inhibition to impact renal physiology. Because the drug stays active in the bloodstream for an extended period due to its long half-life, its effects on renal regulation are continuous, necessitating diligent monitoring of kidney function at baseline, 1-2 weeks post-initiation, and monthly for the first two months. For many patients, the impact of standard 7.5 mg to 15 mg daily dosing is subtle, yet it can trigger a cascade of physiological changes.
The mechanism behind this influence is rooted in the inhibition of essential renal prostaglandins. As noted by the FDA prescribing information, “when these prostaglandins are inhibited, afferent-arterial tone increases, renal blood flow and GFR fall, sodium and water are retained, and the resultant volume expansion and heightened renal-vascular resistance stimulate the renin-angiotensin-aldosterone system and sympathetic activity, producing an elevation in systemic vascular resistance and blood pressure.” In susceptible individuals, this can result in a rapid decline in glomerular filtration rate (GFR) over mere hours or days.
Patients often interpret minor pedal edema—a ‘little bit of swelling’—as a trivial side effect of anti-inflammatory therapy. However, clinically, this is a vital red flag. It serves as a physical signal that the body is retaining excess sodium and fluid, which directly contributes to higher intravascular volume and, consequently, elevated blood pressure. Given that all non-aspirin NSAIDs carry class-wide Black Box warnings, this localized swelling should be the immediate impetus for a comprehensive blood pressure check. While recent real-world evidence from 2020-2024 suggests that meloxicam may offer a more favorable profile than naproxen in high-risk patients—evidenced by an adjusted hazard ratio for thromboembolic events of roughly 3 compared to 10 for naproxen in post-MI cohorts—it remains a potent pharmacological agent that demands proactive home blood pressure monitoring to navigate its therapeutic benefits safely.
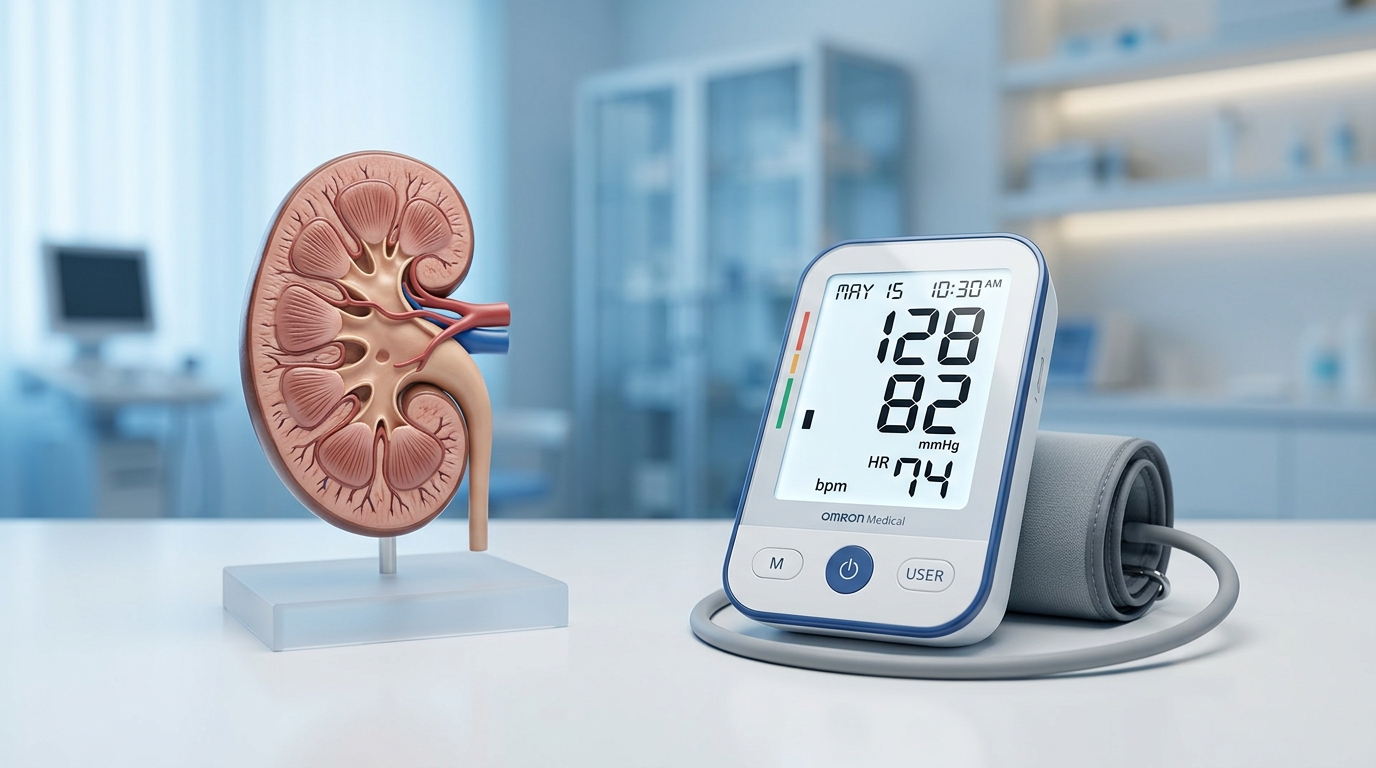
The Renal Filter: Keeping Your Kidneys Safe During Treatment
To understand the renal impact of medications like meloxicam, one must first recognize the kidneys as sophisticated pressure-regulation systems. The filtration process, known as the glomerular filtration rate (GFR), relies on the delicate balance of prostaglandins to keep the afferent arterioles dilated, ensuring that blood can be effectively filtered. Because meloxicam, while preferentially selective for COX-2, still exerts some inhibition on COX-1, it can disrupt this pressure-dependent mechanism. In susceptible patients, this interruption can trigger a rapid fall in GFR within hours or days of treatment initiation.
While meloxicam is often prescribed in standard dosages of 7.5 mg to 15 mg once daily, its long half-life—which provides consistent, long-lasting relief—also means the drug remains active in the bloodstream for an extended period. This makes diligent monitoring essential, particularly for those at higher risk. According to research published in the National Center for Biotechnology Information, “the incidence rate of renal side effects associated with the use of selective and nonselective COX-2 inhibitors is low in otherwise healthy subjects but can get as high as 20% in high risk patients.”
As we integrate current real-world evidence into clinical practice, the emphasis on patient safety has never been higher. To mitigate these risks, the clinical protocol necessitates renal function monitoring at baseline, followed by checks at 1–2 weeks post-initiation, and continuing monthly for the first 1–2 months of therapy. By coupling these routine lab assessments with the future promise of home blood pressure monitoring, providers can better manage the balance between effective pain relief and renal stability. While newer data suggests meloxicam may hold a more favorable safety profile than naproxen in certain high-risk cardiovascular cohorts, the kidneys remain a primary focal point for ongoing medical oversight.
Understanding Your Lab Results: The Role of Creatinine and eGFR
When you undergo a blood panel while taking medication like meloxicam, you are essentially checking the health of your body’s filtration system. For patients prescribed 7.5 mg to 15 mg of meloxicam daily, understanding your renal labs is vital. Because meloxicam is preferentially selective for COX-2—a feature originally marketed to make it more stomach-friendly than older, non-selective NSAIDs—it still maintains some COX-1 inhibition that requires careful oversight. Most importantly, NSAIDs can cause a rapid, often silent, decline in glomerular filtration rate (GFR) over just a few hours or days in vulnerable individuals.
This is why your physician focuses on two key numbers: serum creatinine and eGFR (estimated Glomerular Filtration Rate). Creatinine is a waste product filtered by the kidneys, while eGFR acts as a scorecard for overall kidney efficiency. As noted in clinical guidance, you must monitor renal function (serum creatinine, eGFR) and urine protein-to-creatinine ratio at baseline, 1-2 weeks after initiation, and then monthly for the first 1-2 months. Because meloxicam possesses a long half-life, it remains active in your bloodstream for an extended period, offering consistent relief while necessitating this proactive monitoring schedule.
While the FDA maintains class-wide Black Box warnings for all non-aspirin NSAIDs, emerging evidence from 2020-2024 offers a more nuanced look at cardiovascular safety. In a large Korean cohort study, the adjusted hazard ratio for thromboembolic events was roughly 3 for meloxicam, significantly lower than the 10 observed with naproxen in post-MI patients. By tracking your labs regularly, you and your doctor can ensure this therapy remains safe, as we move toward a future of personalized NSAID dosing based on your unique genetic metabolism and real-time blood pressure data.
Who Is Most at Risk? Identifying Key Vulnerabilities
While meloxicam was historically marketed as a gastrointestinal-sparing alternative to traditional NSAIDs, clinical vigilance remains paramount for specific patient populations. The medication’s preferential selectivity for COX-2 enzymes, while providing a degree of gastric safety, still involves some COX-1 inhibition, necessitating a cautious approach for the elderly, those with diabetic complications, and individuals living with heart failure. Given that NSAIDs can trigger a rapid decline in glomerular filtration rate (GFR) within hours of administration, renal function monitoring is non-negotiable; clinicians should establish a baseline, re-evaluate one to two weeks post-initiation, and maintain monthly check-ins throughout the first two months of therapy.
Cardiovascular risk management has evolved significantly in recent years. While the FDA maintains class-wide Black Box warnings for all non-aspirin NSAIDs, emerging real-world evidence suggests that meloxicam may offer a more favorable profile than other agents in patients with a history of myocardial infarction. As noted in research published by The Hospitalist, some therapies, “including naproxen, conferred a significantly higher risk for cardiovascular and bleeding events, compared with celecoxib and meloxicam.” Supporting this, data from a large Korean cohort indicated an adjusted hazard ratio for thromboembolic events of approximately 3 for meloxicam, notably lower than the 10 observed with naproxen.
For these high-risk profiles, standard dosing—ranging from 7.5 mg to 15 mg once daily—should be approached with extreme discretion. Because meloxicam possesses a long half-life, the drug remains active in the bloodstream for an extended duration; while this ensures consistent therapeutic relief, it also means that adverse systemic effects can linger. As we look toward the future, the integration of genetic markers for drug metabolism and the expansion of home blood pressure monitoring will likely become standard practice, allowing us to tailor dosage more precisely to the individual rather than relying on a one-size-fits-all protocol.
The Proactive Patient: Best Practices for Long-Term Safety
While meloxicam was originally marketed as a more gastrointestinal-friendly alternative to traditional NSAIDs, its long half-life—which provides consistent, steady relief—also necessitates a disciplined approach to long-term usage. As discussed in our Meloxicam 101: Your Daily Guide to Steady Joint Relief, maintaining the standard 7.5 mg to 15 mg once-daily dosage is essential to managing risks. Emerging real-world evidence from 2020 to 2024 suggests that meloxicam’s preferential COX-2 selectivity may offer a more favorable cardiovascular profile compared to naproxen, particularly for those with prior heart conditions. However, patients must remain vigilant; because NSAIDs can trigger a rapid decline in glomerular filtration rate (GFR) within hours or days, proactive monitoring is non-negotiable.
Renal health remains the cornerstone of safety for any long-term anti-inflammatory regimen. Patients should adhere to a strict monitoring schedule: a baseline evaluation, follow-up testing at 1–2 weeks post-initiation, and monthly labs for the first two months. Beyond clinical testing, your daily habits play a vital role. Consistent hydration is essential to support kidney perfusion, and incorporating home blood pressure monitoring can provide your physician with critical real-time data between appointments. According to recent clinical reports on NSAID usage, patients must be hyper-aware of systemic changes. “Early signs may include fatigue, swelling in the hands or feet, changes in urination, and nausea.” If you notice any of these symptoms, contact your healthcare provider immediately to adjust your therapeutic strategy. As we look toward a future defined by personalized medicine and genetic screening for drug metabolism, the most effective tool in your kit remains your own awareness and partnership with your clinical team.