Meloxicam and Your Stomach: Understanding the Risks
By Frankie Sze | 5/1/2026
The GI Dilemma: Why NSAIDs and Stomachs Clash
For decades, traditional non-steroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen and naproxen have served as the frontline against chronic pain, though their reputation for inducing gastric ulcers often precedes them. As noted in recent clinical research, “Both medications reduce inflammation by blocking cyclooxygenase enzymes that produce prostaglandins, the compounds responsible for pain, swelling, and fever.” By suppressing these enzymes, these drugs inadvertently dismantle the stomach’s natural defense mechanisms—specifically, the production of the protective mucus layer that shields the gastric lining from acidic erosion.
In the present landscape of pain management, meloxicam has become a widely prescribed alternative for chronic arthritis, favored for its once-daily dosing regimen of 7.5 mg to 15 mg and a more favorable, albeit imperfect, gastrointestinal safety profile. In fact, comparative data shows that GI adverse events occurred in 30.3% of meloxicam users compared to 44.7% of naproxen users during a six-month trial. Patients seeking to minimize discomfort can often reduce gastric irritation by 40-60% simply by taking meloxicam with food, a practical step detailed further in our guide, Meloxicam 101: Your Daily Guide to Steady Joint Relief.
While clinicians appreciate that meloxicam is metabolized by CYP2C9—avoiding the significant drug interactions that can occur with omeprazole, which is metabolized by CYP2C19 and CYP3A4—patients must remain vigilant. Combining two NSAIDs significantly amplifies the risk of stomach ulcers, GI bleeding, and renal injury, and the medication must be avoided entirely after 20 weeks of pregnancy due to the risk of oligohydramnios. Looking ahead, ongoing monitoring for cardiovascular and kidney health remains an essential pillar of long-term management for those utilizing these therapies for degenerative conditions.
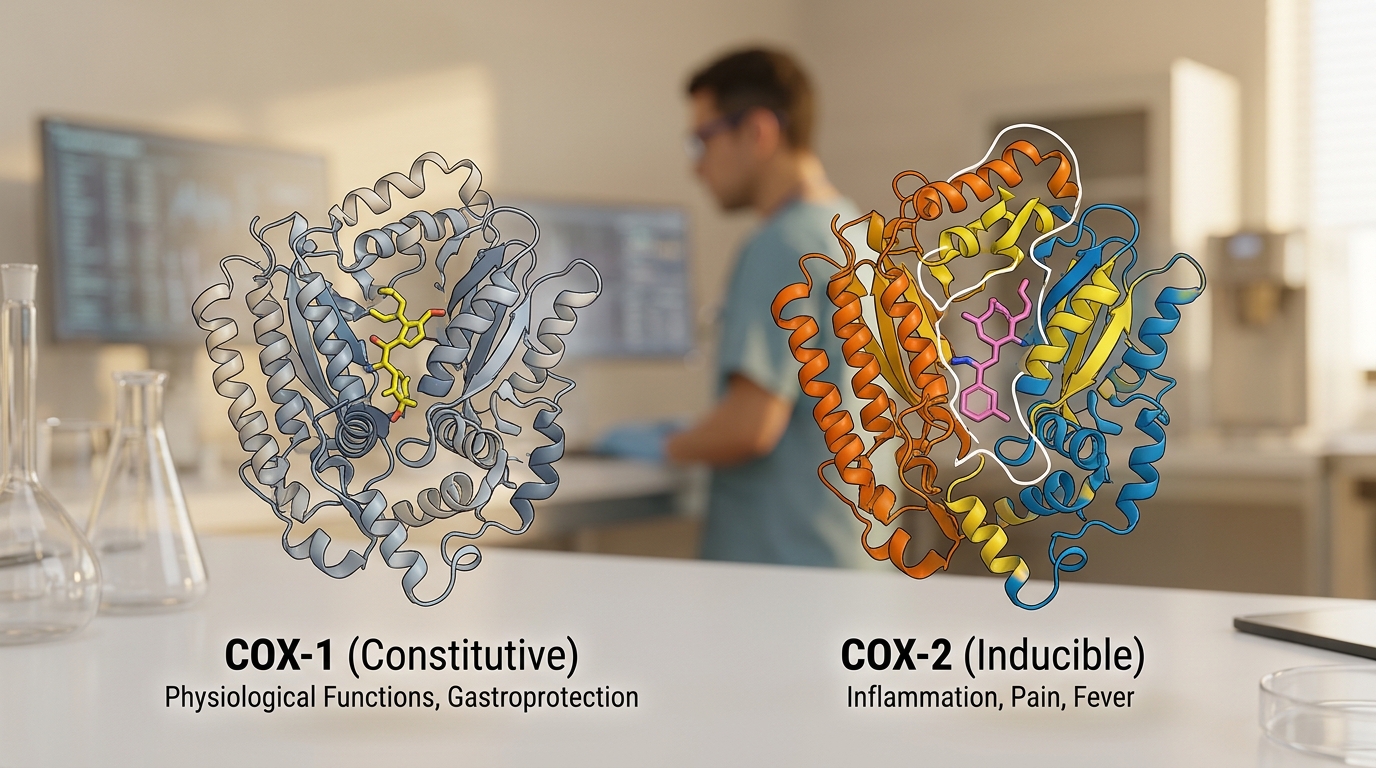
Mechanism Spotlight: How COX-2 Selectivity Protects the Gut
Historically, traditional non-steroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen and naproxen have served as the clinical standard for pain management, yet their development as non-selective inhibitors frequently resulted in high rates of gastric ulcers. This occurs because non-selective agents suppress both the inducible COX-2 enzyme—which drives inflammation—and the constitutive COX-1 enzyme, which plays a critical role in maintaining the protective gastric mucosal lining. As noted in clinical literature, meloxicam is preferentially selective for COX-2 but also inhibits COX-1, a nuance that explains its improved, though not perfect, gastrointestinal safety profile compared to its predecessors.
In practice, this preferential inhibition manifests in tangible patient outcomes; a six-month trial demonstrated that GI adverse events occurred in 30.3% of meloxicam users, significantly lower than the 44.7% observed in those taking naproxen. Meloxicam’s once-daily dosing regimen of 7.5 mg to 15 mg has made it a cornerstone of chronic arthritis management, though patients are encouraged to take the medication with food, which reduces gastric irritation by 40-60%. For those managing complex regimens, clinical pharmacokinetics offer further reassurance: meloxicam is metabolized by CYP2C9, while common gastroprotective agents like omeprazole utilize the CYP2C19 and CYP3A4 pathways, resulting in no significant drug interaction.
Despite these improvements, vigilance remains a clinical imperative. Combining meloxicam with other NSAIDs is strictly contraindicated, as it substantially elevates the risk of stomach ulcers, GI bleeding, and acute kidney injury. Furthermore, clinicians must maintain awareness of broader systemic risks; NSAIDs should be strictly avoided after 20 weeks of pregnancy due to the risk of reduced amniotic fluid. As we look to the future, ongoing monitoring for cardiovascular and kidney health remains essential for patients relying on meloxicam for long-term management of degenerative disorders.
Decoding the Risk: Ulcers and Bleeding Potential
Historically, traditional non-selective NSAIDs like ibuprofen and naproxen have been associated with significant rates of gastric ulceration, casting a long shadow over long-term pain management. In contrast, meloxicam has become a widely prescribed alternative for chronic arthritis, favored for its convenient once-daily dosing—typically ranging from 7.5 mg to 15 mg—and a more favorable gastrointestinal safety profile. However, this improved safety does not equate to an absence of risk.
Clinical data underscores the distinction between these therapeutic classes; in a six-month trial, only 30.3% of those taking meloxicam had gastrointestinal problems, while 44.7% of naproxen users experienced such issues. While this represents a clear reduction in risk, patients must remain vigilant. Practical mitigation strategies, such as taking meloxicam with food, can further reduce the risk of gastric irritation by 40-60%. Furthermore, for patients requiring acid suppression, meloxicam offers a favorable profile: it is metabolized by CYP2C9, while omeprazole is processed by CYP2C19 and CYP3A4, resulting in no significant drug interaction.
Clinical caution is mandatory beyond simple dosage management. Patients should be aware that combining two NSAIDs simultaneously significantly amplifies the risks of stomach ulcers, gastrointestinal bleeding, and renal impairment. Furthermore, the clinical mandate remains firm: NSAIDs should be strictly avoided after 20 weeks of pregnancy due to risks such as reduced amniotic fluid (oligohydramnios). As we look toward the future of chronic care, ongoing monitoring for cardiovascular and kidney health remains an essential component of long-term meloxicam management for degenerative disorders.

Red Flags: Recognizing Early Signs of Stomach Distress
While meloxicam offers a more favorable gastrointestinal safety profile compared to traditional non-selective inhibitors like naproxen—which historically induced ulcers in nearly 45% of users in comparative trials—patients must remain vigilant. Despite meloxicam causing GI adverse events in approximately 30.3% of users, the risk of serious complications remains, particularly when safety protocols are ignored. Patients are advised that taking medication with food can reduce gastric irritation by 40% to 60%, yet this does not eliminate the risk of deeper pathology. It is critical to avoid combining meloxicam with other NSAIDs, as this compound practice exponentially increases the likelihood of stomach ulcers, gastrointestinal bleeding, and renal impairment. Furthermore, those who are pregnant should discontinue NSAID use after 20 weeks due to risks such as reduced amniotic fluid, which can jeopardize fetal development. As noted by SingleCare, patients must seek immediate medical attention if they experience signs or symptoms of an allergic reaction, such as a severe rash, hives, difficulty breathing, or swelling of the face and throat. Any sign of persistent black or tarry stools, coffee-ground emesis, or localized abdominal pain that does not resolve requires urgent evaluation, as these may be harbingers of severe GI distress requiring immediate clinical intervention.
Best Practices: Habits to Minimize Gastric Irritation
As clinical practice shifts from the non-selective NSAIDs of the past—which were notorious for high rates of gastric ulceration—toward targeted therapies like meloxicam, patient safety remains centered on managing digestive tolerance. Meloxicam has become a staple for chronic arthritis management, favored for its convenient once-daily dosing at 7.5 mg to 15 mg and a significantly improved gastrointestinal safety profile, with trials showing GI adverse events in 30.3% of users compared to 44.7% for those on naproxen. However, vigilance is still required, as long-term use for degenerative conditions necessitates ongoing monitoring for cardiovascular and renal risks.
The most accessible strategy for mitigating sensitivity is dietary modification. According to research from Doctronic, “Taking meloxicam with food reduces stomach upset by 40-60% without meaningfully impacting the medication’s pain-relieving effectiveness.” For patients requiring additional protection, such as those on long-term anti-inflammatory therapy, proton pump inhibitors (PPIs) like omeprazole may be recommended. Because meloxicam is metabolized via the CYP2C9 pathway while omeprazole primarily utilizes CYP2C19 and CYP3A4, clinicians can confidently prescribe them in tandem, as there is no significant pharmacokinetic interaction between these two classes.
Patients must be cautioned against the common pitfall of combining multiple NSAIDs, which exponentially increases the risk of stomach ulcers, gastrointestinal bleeding, and kidney damage. Furthermore, specific life stages demand strict avoidance; for instance, NSAIDs should not be used after 20 weeks of pregnancy due to the risk of reduced amniotic fluid levels. By adhering to these evidence-based habits—pairing medication with food, avoiding multi-drug NSAID regimens, and maintaining appropriate clinical oversight—patients can better navigate the balance between effective pain management and long-term digestive health.
The ‘At-Risk’ Profile: Who Should Take Extra Precautions
While meloxicam is widely prescribed for chronic arthritis due to its once-daily 7.5 mg to 15 mg dosing and improved gastrointestinal safety profile compared to older, non-selective NSAIDs like ibuprofen or naproxen, it is not without risk. For patients navigating long-term pain management, understanding the specific contraindications is vital. A six-month trial demonstrated that GI adverse events occurred in 30.3% of meloxicam users compared to 44.7% of those on naproxen; however, individual risk factors can alter this safety balance significantly.
Pregnant women represent a high-risk group that requires strict vigilance. As noted by experts at SingleCare, “NSAIDs should be avoided in pregnant women at around 20 weeks or later in pregnancy” due to the potential for serious complications, including reduced amniotic fluid levels. Furthermore, patients currently taking multiple medications should exercise extreme caution; combining meloxicam with another NSAID dramatically increases the risk of stomach ulcers, GI bleeding, and kidney damage. While it is reassuring that meloxicam—which is metabolized by CYP2C9—does not have significant drug interactions with omeprazole, which utilizes different metabolic pathways, patients should still seek comprehensive guidance through resources like Meloxicam 101: Your Daily Guide to Steady Joint Relief to ensure their medication regimen is optimized.
For those managing chronic conditions, simple behavioral modifications can provide an added layer of safety. Taking meloxicam with food has been shown to reduce gastric irritation by 40-60%. As we look to the future, ongoing clinical monitoring for cardiovascular and kidney health remains the gold standard for patients using these therapies for the long-term management of degenerative disorders. Frankie Sze suggests that patients maintain a transparent dialogue with their healthcare providers regarding all concurrent supplements and medications to mitigate avoidable risks.