Meloxicam vs. Ibuprofen: Comparing Stomach Safety and GI Risks
By Frankie Sze | 5/2/2026
Introduction: The GI Dilemma in Chronic Pain Management
For decades, traditional non-steroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen and naproxen served as the frontline defense against chronic joint pain. However, their clinical utility was historically curtailed by a persistent conflict: the need for sustained anti-inflammatory relief versus the significant risk of gastric mucosal damage. As patients often ask, common safety questions include which drug has a lower risk of stomach irritation, reflecting the ongoing struggle to balance effective pain management with patient safety.
This pursuit of a therapeutic middle ground led to the development of COX-2 selective inhibitors. Meloxicam has emerged as a leading prescription choice for chronic arthritis, favored for its potency and convenient once-daily dosing of 7.5 mg to 15 mg, a marked improvement over the frequent every 4-6 hour dosing required by ibuprofen. Meloxicam demonstrates a COX-2 selectivity ratio of approximately 18, meaning it requires 18 times the concentration to inhibit COX-1—the enzyme responsible for protecting the stomach lining—as it does COX-2. This biochemical profile translates to clinical outcomes: meloxicam reduces the risk of ulcer complications by approximately 47 percent compared to non-selective NSAIDs.
Despite these advancements, risks remain. Serious gastrointestinal complications occur in 2 percent of meloxicam patients annually, compared to 3.5 percent for ibuprofen patients. These risks are notably higher in elderly populations (60+), where the likelihood of upper-GI complications reaches roughly 1 percent after 3-6 months and 2-4 percent after one year of use. For those navigating these concerns, understanding the nuances of safe administration is critical, as detailed in our guide, Meloxicam 101: Your Daily Guide to Steady Joint Relief. Today, gastroprotective strategies have evolved, with PPI co-therapy now capable of reducing NSAID-related upper-GI ulcers by approximately 90 percent. As we look to the future, clinical practice is shifting toward fixed-dose combinations of NSAIDs and PPIs to improve adherence, while clinicians continue to monitor the long-term cardiovascular profiles of all COX-2 selective agents to ensure patient wellbeing remains the priority.
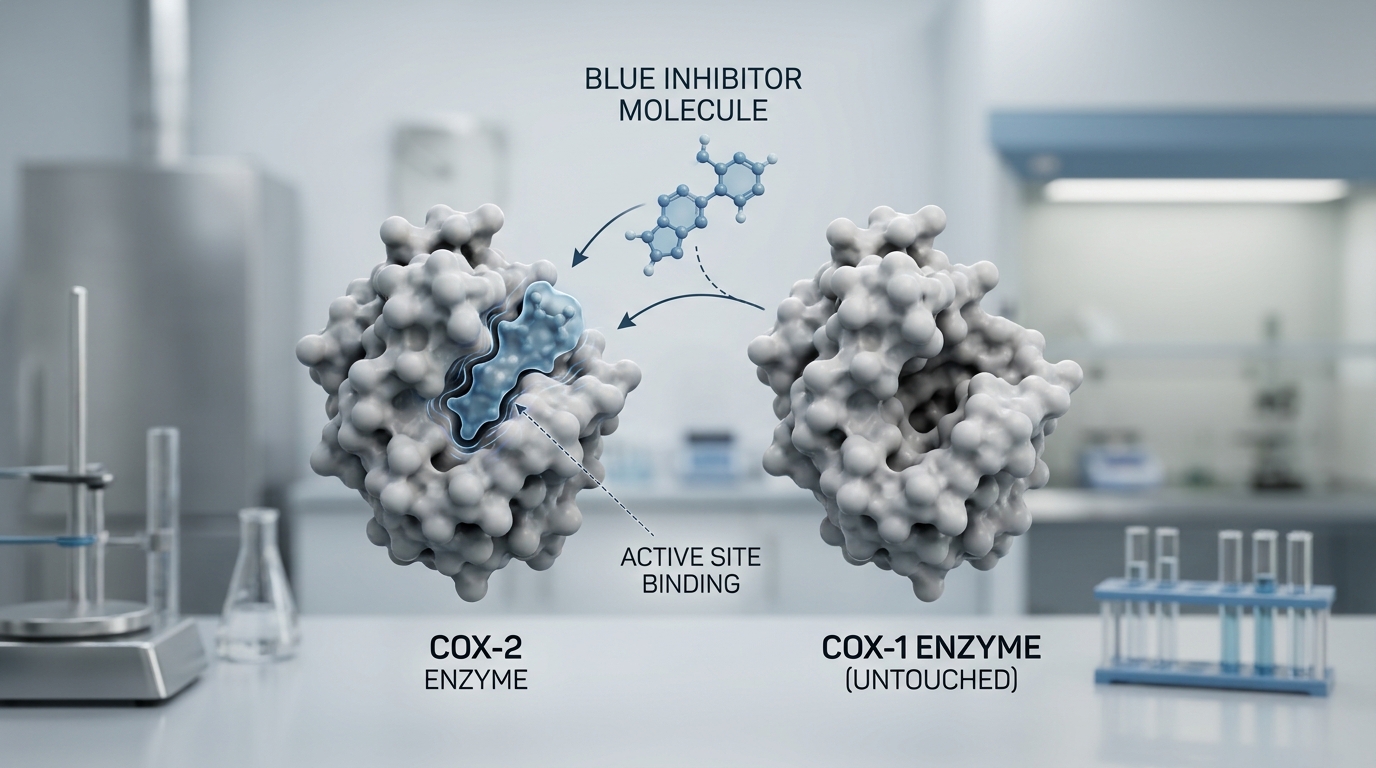
COX-1 vs. COX-2: Understanding the Biological ‘Shield’ of the Stomach
To understand the clinical evolution of non-steroidal anti-inflammatory drugs (NSAIDs), one must first grasp the duality of the cyclooxygenase (COX) enzymes. As noted by SingleCare, “Ibuprofen is a non-selective NSAID that blocks both COX-1 and COX-2 enzymes.” While COX-2 is primarily associated with the inflammation and pain experienced in chronic conditions like arthritis, COX-1 serves a vital, constitutive role in maintaining the gastric mucosal lining. Traditional NSAIDs, by indiscriminately inhibiting both enzymes, inadvertently strip away this biological shield, leading to the high rates of gastric ulcers that have historically limited their long-term use.
The therapeutic shift toward COX-2 selectivity represents a significant advancement in gastrointestinal safety. Meloxicam serves as a prime example of this balance, featuring a COX-2 selectivity ratio of approximately 18, which implies it requires 18 times the concentration to inhibit the stomach-protecting COX-1 enzyme compared to the inflammatory COX-2. This biochemical specificity translates into tangible clinical benefits: patients utilizing meloxicam experience serious GI complications at an annual rate of 2 percent, compared to 3.5 percent for those on ibuprofen. Furthermore, meloxicam reduces the overall risk of ulcer complications by approximately 47 percent when contrasted with non-selective alternatives.
Clinical management of chronic pain now emphasizes this tolerability, though vigilance remains essential. For elderly patients (60+), the risk of upper-GI complications is roughly 1 percent after 3-6 months of meloxicam use, climbing to 2-4 percent after one year. To mitigate these risks further, current best practices often involve PPI co-therapy, which can reduce NSAID-related upper-GI ulcers by about 90 percent. Unlike ibuprofen, which mandates frequent dosing every 4-6 hours, meloxicam’s pharmacokinetics allow for once-daily dosing at 7.5 mg to 15 mg, offering both enhanced gastrointestinal safety and improved patient adherence in the treatment of chronic arthritic conditions.
The Meloxicam Mechanism: How Selective Inhibition Reduces Irritation
For decades, traditional NSAIDs such as ibuprofen and naproxen have served as the standard-bearer for pain management, yet their clinical utility was perpetually constrained by high rates of gastric ulceration and systemic irritation. The pharmacological evolution toward COX-2 selective inhibitors was driven by the urgent need to dissociate anti-inflammatory efficacy from the deleterious effects on the gastric mucosa. As noted by research experts, “Meloxicam targets the COX-2 enzyme, reducing inflammation and pain with fewer stomach side effects than many traditional NSAIDs.”
This clinical advantage is rooted in meloxicam’s precise biochemical profile. It maintains a COX-2 selectivity ratio of approximately 18, meaning the drug requires a concentration 18 times higher to inhibit the constitutive, cytoprotective COX-1 enzyme compared to the inducible, pro-inflammatory COX-2 enzyme. This preferential inhibition results in a tangible safety advantage: while serious gastrointestinal (GI) complications occur in roughly 3.5 percent of ibuprofen patients, that figure drops to approximately 2 percent for those prescribed meloxicam. Consequently, the drug reduces the risk of ulcer-related complications by nearly 47 percent when compared to non-selective alternatives.
The impact of this mechanism is particularly significant for vulnerable populations. In elderly patients aged 60 and older, the risk of upper-GI complications remains manageable, measuring roughly 1 percent after 3-6 months of use, and scaling to 2-4 percent over a one-year duration. Clinicians are further refining these outcomes through standardized gastroprotective protocols; for instance, concurrent PPI co-therapy has been shown to reduce NSAID-related upper-GI ulcers by approximately 90 percent. Today, meloxicam has emerged as a premier prescription choice for chronic arthritis, balancing potent anti-inflammatory action with the convenience of a once-daily 7.5 mg to 15 mg dose, a stark contrast to the repetitive 4-6 hour dosing schedule required by traditional agents like ibuprofen. As we look toward the future, the medical community remains focused on the long-term monitoring of cardiovascular markers and the integration of fixed-dose NSAID-PPI combinations to ensure both patient comfort and therapeutic adherence.
Data Deep Dive: GI Complication Rates of Meloxicam vs. Ibuprofen and Naproxen
For decades, traditional non-selective nonsteroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen and naproxen served as the frontline defense against arthritis-related pain. While efficacious, their therapeutic utility was frequently curtailed by significant gastrointestinal (GI) toxicity resulting from the non-selective inhibition of both COX-1 and COX-2 enzymes. The evolution of pharmacotherapy shifted toward agents with higher selectivity, such as meloxicam, which possesses a COX-2 selectivity ratio of approximately 18—meaning it requires 18 times the concentration to inhibit COX-1 compared to its target, COX-2. This biochemical profile is central to its improved tolerability.
Clinical data underscores this safety advantage, as serious gastrointestinal complications (bleeding, perforation or stricture) occur in roughly 2 percent of patients treated with meloxicam each year compared with about 3.5 percent of patients receiving ibuprofen. By minimizing impact on protective gastric prostaglandins, meloxicam reduces the overall risk of ulcer complications by approximately 47 percent compared to non-selective counterparts. This is particularly relevant for the vulnerable elderly population (aged 60+), where the risk of upper-GI complications with meloxicam remains controlled at roughly 1 percent after 3-6 months of use, and 2-4 percent after one year.
Beyond the inherent pharmacological profile, patient adherence plays a critical role in GI health. Meloxicam is typically prescribed as a once-daily dose of 7.5 mg to 15 mg, whereas ibuprofen often necessitates dosing every 4-6 hours, which can complicate therapeutic regimens. Furthermore, current clinical standards emphasize the role of gastroprotection; the integration of proton pump inhibitor (PPI) co-therapy has been shown to reduce NSAID-related upper-GI ulcers by approximately 90 percent. Looking ahead, while we anticipate the continued adoption of fixed-dose combinations of NSAIDs and PPIs to bolster patient safety, clinicians remain focused on the long-term cardiovascular monitoring of all COX-2 selective agents to ensure that the systemic benefits of chronic pain management remain balanced with patient safety profiles.
Residual Risks: Why ‘Gentler’ Doesn’t Mean ‘Risk-Free’
For decades, traditional non-steroidal anti-inflammatory drugs (NSAIDs) like ibuprofen and naproxen served as the frontline defense against arthritis pain, yet their utility was frequently undermined by significant gastric toxicity. The introduction of agents like meloxicam represented a shift toward COX-2 selective inhibition, designed to offer anti-inflammatory relief while minimizing damage to the stomach lining. With a COX-2 selectivity ratio of approximately 18—meaning it requires 18 times the concentration to inhibit COX-1 compared to COX-2—meloxicam has become a leading prescription choice, favored for its once-daily 7.5 mg to 15 mg dosing schedule, which stands in stark contrast to the frequent 4-6 hour dosing required by ibuprofen.
However, this perceived gentleness is not synonymous with an absence of risk. As experts note, “meloxicam’s COX-2 selectivity tends to cause fewer gastrointestinal side effects but still carries ulcer and bleeding risk.” While meloxicam reduces the risk of ulcer complications by roughly 47 percent compared to non-selective NSAIDs, it remains a potent medication. Clinical data bears this out: serious gastrointestinal complications occur in 2 percent of meloxicam patients annually. This figure is particularly critical for vulnerable populations; in patients aged 60 and older, the risk of upper-GI complications climbs to approximately 1 percent after three to six months of use, reaching 2 to 4 percent after one full year.
Recognizing these residual dangers is vital for long-term management. We are seeing a future shift toward fixed-dose combinations and mandatory protective protocols, as the concurrent use of proton pump inhibitors (PPIs) can reduce NSAID-related upper-GI ulcers by approximately 90 percent. As we continue to monitor the long-term cardiovascular profile of all COX-2 selective agents, patients must remember that convenience should never be mistaken for clinical immunity. For those relying on meloxicam for chronic arthritis, active management of these lingering risks is the final pillar of responsible care.
Protective Strategies: Food, Timing, and the Role of PPIs
While meloxicam represents a significant clinical improvement over traditional non-selective NSAIDs—boasting a COX-2 selectivity ratio of approximately 18—it remains a potent pharmacological agent that requires proactive gastrointestinal management. Historically, agents like ibuprofen were the standard for arthritis pain; however, they required frequent dosing every 4–6 hours, which contributed to a 3.5 percent annual rate of serious GI complications. In contrast, meloxicam’s once-daily dosing (7.5 mg to 15 mg) simplifies patient adherence while reducing the risk of ulcer complications by roughly 47 percent compared to non-selective alternatives. Despite this improved profile, the annual risk of serious GI events remains at 2 percent, particularly climbing in elderly patients (60+) to 1–4 percent over the course of a year.
To mitigate these risks, taking meloxicam with food or a full glass of water remains a foundational recommendation. However, lifestyle adjustments alone are often insufficient for long-term users. Clinical data underscores the necessity of pharmacological protection for those at elevated risk. As noted by experts, “PPIs reduce the risk of NSAID-related ulcers by approximately 90% and are the only gastroprotective strategy with robust evidence in this high-risk scenario” (DrOracle Gastroprotective Guidelines). By integrating proton pump inhibitor (PPI) co-therapy, patients can achieve a nearly complete reduction in NSAID-induced upper-GI ulceration. As we look toward future care models, the medical community is moving toward fixed-dose combinations of NSAIDs and PPIs to ensure this protective standard is maintained consistently across chronic treatment plans.
Patient Profile: Who Benefits Most from Meloxicam’s Stomach-Sparing Profile?
For patients managing chronic arthritis, the legacy of traditional NSAIDs like ibuprofen and naproxen is often defined by a difficult trade-off: effective pain relief at the expense of gastric health. Meloxicam has emerged as a cornerstone of modern rheumatological care by addressing this historical limitation. With a COX-2 selectivity ratio of approximately 18, meloxicam targets inflammation with greater precision, requiring 18 times the concentration to inhibit the protective COX-1 enzymes that line the stomach compared to its effect on COX-2. As noted by The Freedom Center, “Meloxicam offers once-daily dosing with longer-lasting effects, making it preferred for chronic inflammatory conditions like arthritis.”
The clinical data underscores a clear benefit for those sensitive to OTC NSAIDs. While ibuprofen requires frequent dosing every 4-6 hours, meloxicam’s once-daily regimen—typically 7.5 mg to 15 mg—improves adherence while significantly reducing gastric stress. Research indicates that serious GI complications occur in only 2 percent of meloxicam patients annually, compared to 3.5 percent for those using ibuprofen. Overall, meloxicam reduces the risk of ulcer complications by approximately 47 percent when compared to non-selective NSAIDs. For elderly patients (60+), the risk remains manageable, measuring roughly 1 percent after 3-6 months and 2-4 percent after one year of consistent use. For those transitioning from shorter-acting medications, Meloxicam 101: Your Daily Guide to Steady Joint Relief provides the necessary roadmap for navigating this therapeutic shift.
Looking ahead, the clinical focus is shifting toward even greater safeguards. While we anticipate ongoing monitoring of long-term cardiovascular risks associated with all COX-2 selective agents, the future of treatment increasingly emphasizes combined protocols. Specifically, the integration of PPI co-therapy has been shown to reduce NSAID-related upper-GI ulcers by approximately 90 percent, a strategy that is becoming standard practice. For the chronic patient, meloxicam represents a vital evolution: a treatment that balances the necessity of potent anti-inflammatory relief with the imperative of long-term gastrointestinal integrity. – Frankie Sze.