Is Meloxicam a Narcotic? Understanding Your Prescription
By Frankie Sze | 5/1/2026
The Prescription Paradox: Why Patients Fear New Painkillers
In the shadow of the opioid crisis, a profound shift has occurred in how patients approach pain management. For many, the sight of a prescription bottle—regardless of the medication class—triggers a reflexive sense of apprehension. This skepticism is a natural, albeit sometimes counterproductive, response to the systemic over-prescribing that characterized the last two decades. As we navigate this landscape, it is vital to distinguish between powerful analgesics that alter the brain’s reward pathways and therapies designed for physiological relief. While clinicians now prioritize safer, long-term options, patients remain wary of the word ‘addictive.’ To help you understand your treatment plan, Meloxicam 101: Your Daily Guide to Steady Joint Relief provides the foundational knowledge necessary to separate fact from fear.
FDA-approved as a long-acting NSAID, meloxicam has emerged as a cornerstone of modern care for osteoarthritis and rheumatoid arthritis. Unlike opioids, meloxicam is not listed under any of the five schedules of the Controlled Substances Act, meaning it carries no inherent addictive potential. It functions by inhibiting COX-1 and COX-2 enzymes rather than interacting with central nervous system reward pathways. Despite its status as one of the most frequently prescribed non-opioid treatments today, some confusion persists regarding its cessation. As noted by Align Sonoma, ‘In the United States alone, over 30 million adults are prescribed NSAIDs each year for chronic pain management, including meloxicam. While meloxicam is generally safe when used as directed, some people who stop taking it may experience withdrawal-like symptoms if they have used it for an extended period.’
It is essential to clarify that ‘rebound inflammation’—a return of stiffness, swelling, or redness—is fundamentally different from the autonomic symptoms of opioid withdrawal, such as tremors or nausea. Because meloxicam does not create chemical dependency, standard oral doses of 7.5 mg to 15 mg once daily can be stopped without a tapering period. Moving forward, as the medical community doubles down on non-opioid alternatives, ongoing education will be the key to ensuring that patients no longer conflate ‘prescription-strength’ with the potential for abuse, keeping safe options like meloxicam at the forefront of orthopedic care.

Defining the Class: What Meloxicam Is (and Isn’t)
In the wake of the national opioid crisis, patients often harbor valid concerns regarding any prescription-strength pain medication. It is critical to establish that meloxicam is not a narcotic; rather, it is a potent Nonsteroidal Anti-Inflammatory Drug (NSAID) FDA-approved for the long-term management of chronic conditions like osteoarthritis and rheumatoid arthritis. As noted by the Aura Recovery Center, “Meloxicam (available as oral tablets, oral capsules, or an oral suspension) is a prescription-only medication classified as a Nonsteroidal Anti-Inflammatory Drug (NSAID) used to alleviate inflammatory pain; it is not a narcotic or an opioid.”
Unlike opioids, which interact with central nervous system reward pathways to produce analgesic effects, meloxicam functions by inhibiting COX-1 and COX-2 enzymes to reduce the production of inflammatory prostaglandins. Because it does not interact with these reward centers, meloxicam is not listed under any of the five schedules of the Controlled Substances Act, nor does it carry the risk of chemical dependency. Patients prescribed the standard dosage of 7.5 mg to 15 mg once daily can cease treatment without the formal tapering period required for narcotics. While discontinuing the medication may lead to a return of original symptoms—a phenomenon characterized by rebound inflammation involving stiffness, swelling, and redness—this process lacks the autonomic physiological symptoms of opioid withdrawal, such as intense nausea or tremors.
As we look to the future of orthopedic care, the prioritization of non-opioid management ensures meloxicam remains a primary therapeutic pillar. Continued clinical education is essential to ensure that patients distinguish between the term “prescription-strength” and the concept of “addictive potential,” helping to alleviate unnecessary stigma surrounding this vital anti-inflammatory treatment.

The Science of the High: Why Meloxicam Can’t Cause Addiction
In the shadow of the opioid crisis, many patients harbor understandable anxiety toward any medication requiring a prescription. However, it is essential to distinguish between the mechanism of narcotics and the pharmacological reality of meloxicam, an FDA-approved non-steroidal anti-inflammatory drug (NSAID) that remains a cornerstone in modern osteoarthritis and rheumatoid arthritis management. Unlike opioids, which interact directly with the central nervous system to induce euphoria and physical dependence, meloxicam exerts its therapeutic effects peripherally. As noted by BetterAddictionCare, “Unlike opioids, meloxicam does not trigger dopamine release in the brain’s prefrontal cortex or limbic reward circuits, which are responsible for addiction-forming reinforcement.”
Because meloxicam functions by inhibiting COX-1 and COX-2 enzymes to reduce inflammation rather than manipulating reward pathways, it is not listed under any of the five schedules of the Controlled Substances Act. This pharmacological profile means that standard oral dosing—typically 7.5 mg to 15 mg once daily—does not lead to chemical dependency. Consequently, patients do not require a tapering period when discontinuing treatment. While some individuals may experience a return of stiffness, swelling, or redness as part of rebound inflammation, this physiological resurgence is distinct from the autonomic symptoms of opioid withdrawal, such as nausea or tremors. As clinical focus shifts toward non-opioid pain management, it is crucial that patients recognize the distinction between ‘prescription-strength’ potency and addictive potential, ensuring that effective, long-term orthopedic care remains accessible without the fear of habituation.
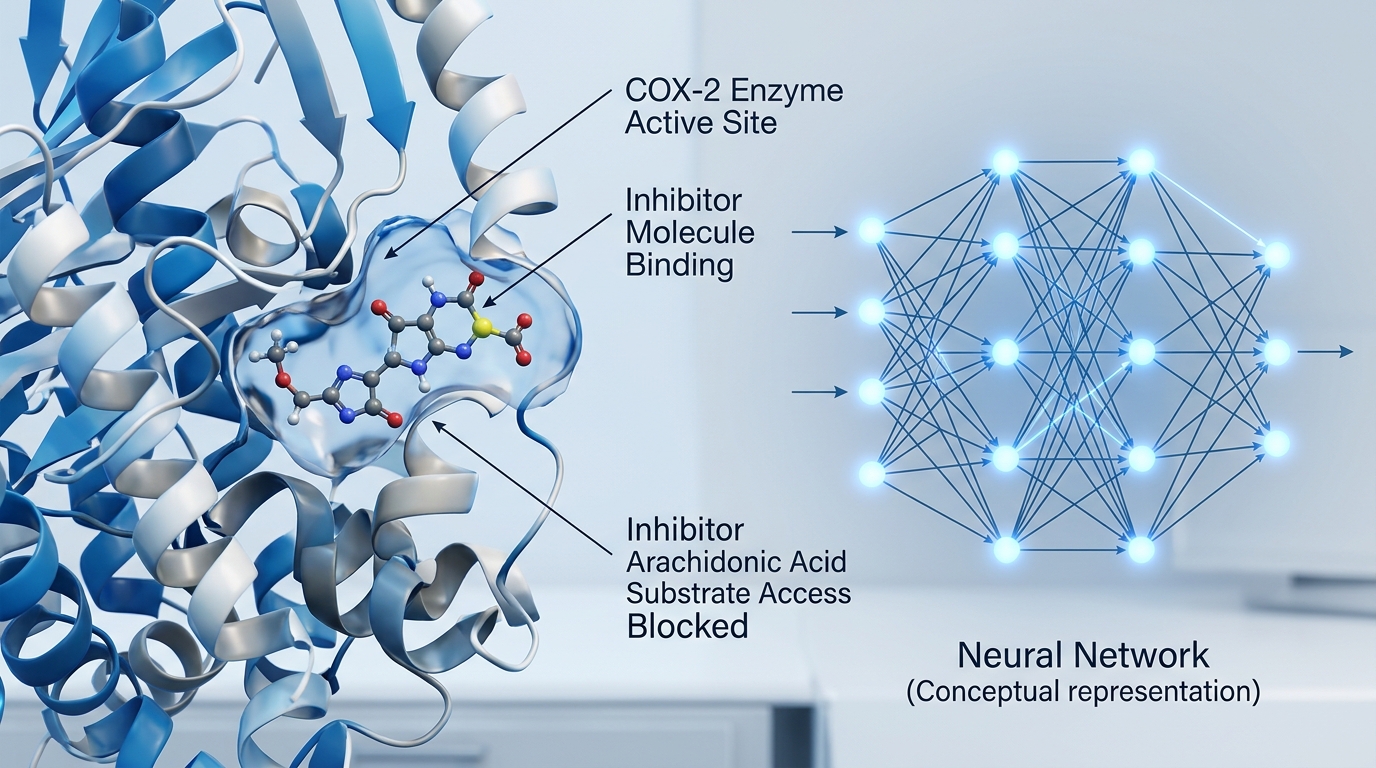
Controlled vs. Non-Controlled: Understanding the DEA Status
In the wake of the national opioid crisis, a significant amount of patient apprehension has emerged regarding the use of prescription-strength pain management. This widespread caution often leads individuals to conflate ‘prescription-only’ status with addictive potential. However, it is essential to clarify that meloxicam is not listed under any of the five schedules of the Controlled Substances Act. As noted by Ascendant NY, ‘No, the U.S. Drug Enforcement Agency (DEA) has not officially classified meloxicam as a controlled substance. While some people assume meloxicam is a controlled substance, it is still classified as a non-psychoactive NSAID.’
The clinical distinction lies in the drug’s mechanism of action: meloxicam works by inhibiting COX-1 and COX-2 enzymes to reduce prostaglandins, rather than by acting on the central nervous system reward pathways that characterize habit-forming analgesics. FDA-approved to provide a safer alternative for chronic arthritis management, meloxicam is now one of the most frequently prescribed non-opioid treatments for osteoarthritis and rheumatoid arthritis. Standard oral dosing typically ranges from 7.5 mg to 15 mg once daily, and because the drug does not induce chemical dependency, patients do not require a formal tapering period when discontinuing treatment. While some patients may experience rebound inflammation—a return of stiffness, swelling, and redness—this response is fundamentally different from opioid withdrawal, as it lacks autonomic symptoms such as nausea or tremors. As the medical community increases its focus on non-opioid pain management, ongoing education remains vital to ensuring patients distinguish between therapeutic necessity and the risks associated with controlled substances.
Withdrawal vs. Rebound: What Happens When You Stop?
In the wake of the national opioid crisis, patients frequently harbor a lingering fear that any ‘prescription-only’ medication carries a high risk of chemical dependency. However, it is essential to distinguish between the clinical profile of opioids and that of meloxicam, a non-steroidal anti-inflammatory drug (NSAID) currently acting as a cornerstone of modern osteoarthritis and rheumatoid arthritis management. Unlike opioids, which interact with central nervous system reward pathways, meloxicam functions by inhibiting COX-1 and COX-2 enzymes to reduce prostaglandins. Because it does not act on these neural pathways, meloxicam is not listed under any of the five schedules of the Controlled Substances Act, and standard daily doses of 7.5 mg to 15 mg do not lead to physical addiction.
Patients often worry about the physical consequences of cessation, but because meloxicam does not cause chemical dependency, no tapering period is required. While you will not experience the autonomic dysregulation characteristic of narcotic withdrawal—such as tremors, severe nausea, or autonomic nervous system distress—you may encounter a ‘rebound’ effect. As noted by The Recovery Village: “Although meloxicam does not cause a withdrawal syndrome, since it is used to treat pain and inflammation, anyone who suddenly stops taking the medication may experience the return of their pain or inflammation. This return would include the symptoms of inflammation such as stiffness, swelling, redness and heat.” This physiological rebound is a direct reflection of the underlying condition resurfacing rather than a symptom of drug withdrawal.
As we look toward the future of orthopedic care, the focus on non-opioid pain management will likely solidify meloxicam’s role in long-term treatment. Nevertheless, clinical providers must continue to emphasize that prescription-strength efficacy does not equate to addictive potential. By understanding the mechanical difference between pharmacological withdrawal and the return of baseline inflammation, patients can navigate their pain management protocols with confidence and clarity.
Safe Pain Management Without the Narcotic Risk
In an era shadowed by the opioid crisis, patients often approach any prescription-strength medication with understandable apprehension. However, it is essential to distinguish between analgesics that target the central nervous system and those that address systemic inflammation at the source. Unlike controlled substances, meloxicam is not listed under any of the five schedules of the Controlled Substances Act. Its mechanism of action is distinctly non-narcotic; it works by inhibiting COX-1 and COX-2 enzymes to reduce inflammation rather than manipulating reward pathways in the brain. As noted by Ikon Recovery Centers, “Neither meloxicam nor ibuprofen is considered addictive, though both carry risks of serious side effects when misused or taken in excessive amounts.”
Because meloxicam does not induce chemical dependency, patients do not require a clinical tapering period when concluding therapy. While users may experience rebound inflammation—characterized by a return of joint stiffness, redness, or swelling—this response is fundamentally different from the autonomic distress of opioid withdrawal, which often includes severe nausea and tremors. With standard oral dosing typically ranging from 7.5 mg to 15 mg once daily, the medication is designed for steady, long-term management of conditions like osteoarthritis and rheumatoid arthritis. For a comprehensive overview of how to integrate this medication into your daily regimen, see our guide, Meloxicam 101: Your Daily Guide to Steady Joint Relief.
As we look toward the future of orthopedic care, ongoing patient education is vital to ensure that the term “prescription-strength” is no longer conflated with “addictive potential.” By focusing on functional improvement and consistent inflammation control rather than temporary euphoria, meloxicam stands as a cornerstone of safe, long-term healing, ensuring patients remain in control of their own recovery journey.